Research Interests
The general research interests of the laboratory are Neural Control and Biomechanics of Movement. We study how neuromechanical systems with seemingly redundant degrees of freedom are managed by the nervous system to produce purposeful motor behaviors and how the neural control of motor behaviors is affected by injury (spinal cord or peripheral nerve injury, stroke, limb amputation or vision loss).
Personnel
- Director: Boris I. Prilutsky, PhD
- Current and past research scientists and postdocs:
- Mohammadali S. Rahmati, PhD
- Alexander N. Klishko, PhD
- Annette L. Pantall, PhD
- Margarita Bulgakova, PhD
- Emma Hodson-Tole, PhD
- Huub Maas, PhD
- Past PhD Students:
- Zhenxuan (James) Zhang, PhD
- Kyunggeune (Ted) Oh, PhD
- Joshua Jarrell, PhD
- Hangue Park, PhD
- Ricky Mehta, PhD
- Bradley J. Farrell, PhD
- Linda Harley, PhD
- Past Master Students:
- Gregory Phillips, MS
- Zhengqin Fan, MS
- Current graduate students
- Claire Hanson, Biology
- Yusuf Islam, Applied Physiology
- Current undergraduate research assistants:
- Ateendra Subramanian, Neuroscience
- Ashley Nguyen, Biology
Current funding
- NSF/ECCS 2024414 (Jia, Li, Prilutsky) Collaborative Research: NCS-FO: Intelligent Closed-Loop Neural Interface System for Studying Mechanisms of Somatosensory Feedback in Control of Functional and Stable Locomotion, 01/2020-08/2024.
- NIH R01 NS110550 (Frigon, Prilutsky, Rybak) Limb coordination during locomotion before and after spinal cord injury, 02/2020-12/2025.
Current collaborators
-
- Turgay Akay, PhD, Dalhousie University
- Irina N. Beloozerova, PhD, Georgia Tech
- Thomas J. Burkholder, PhD, Georgia Tech
- Timothy C. Cope, PhD, Georgia Tech
- John F. Dalton IV, MD, Georgia Hand & Microsurgery
- Tatiana N. Deliagina, PhD, Karolinska Institute
- Alain Frigon, PhD, Université de Sherbrooke
- Yury Gerasimenko, PhD, DSc, Pavlov Institute of Physiology, University of Louisville
- Robert J. Gregor, PhD, Georgia Tech and University of South California
- Yaoyao Jia, PhD, North Carolina State University
- Mark L. Latash, PhD, Penn State University
- Michel A. Lemay, PhD, Drexel University College of Medicine
- Wen Li, PhD, Michigan State University
- Sergei Markin, PhD, Drexel University College of Medicine
- Pavel E. Musienko, PhD, DSc, St. Petersburg State University, Pavlov Institute of Physiology
- Richard Nichols, PhD, Georgia Tech
- Mark Pitkin, PhD, Tufts University School of Medicine
- Ilya A. Rybak, PhD, Drexel University College of Medicine
Current and Past Research Projects
- Neural mechanisms of locomotion evoked by epidural stimulation of the spinal cord
The overall goal of this project is to determine the contribution of motion-dependent afferent pathways, selected ascending and descending pathways in the spinocerebellar loop, and the central pattern generator (CPG) circuitry to the generation of the distinct kinematic and muscle synergies during normal walking and walking evoked by the epidural spinal cord stimulation (ES) in cats with intact and partially transected spinal cord. This goal will be accomplished in experimental and neuromechanical computational studies performed in close collaboration among 4 research groups: Georgia Institute of Technology (our lab); Karolinska Institute, Sweden; Drexel University and Pavlov Institute of Physiology, Russia. In this study, we use the neuromechanical computational model of spinal control of locomotion (Markin et al. 2016) to interpret the experimentally obtained kinematic and muscle synergies and activity patterns of spinal interneurons during the MLR-evoked and ES-evoked walking in the decerebrate cat.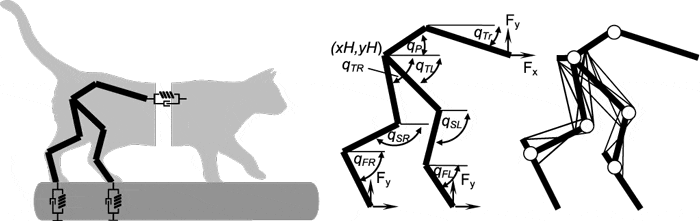
- Sensory Mechanisms of Functional Compensation After Peripheral Nerve Injury
In this project we investigate (1) the effects of lost input from the primary spindle afferents in proximal muscles as a result of peripheral nerve injury and repair on interlimb, interjoint and intermuscle coordination during locomotion and (2) the mechanisms underlying functionally appropriate compensations after partial paralysis of a muscle group. This project involves close collaboration with Drs. Robert J. Gregor (Georgia Tech), Arthur W. English (Emory University), and T. Richard Nichols (Georgia Tech). Spinal circuitry and mechanical consequences of loss of muscle length feedback are simulated in collaboration with Dr. Donald H. Edwards (Georgia State University) using software AnimatLab developed in his group, Dr. Gennady Cymbalyuk (Georgia State University), and Dr. Ilya A. Rybak (Drexel University) using software NSM developed in his group.

- Mobility Skill Acquisition and Learning through Alternative and Multimodal Perception for Visually Impaired People
In this project we study the role of different modes of sensory feedback information (visual, somatosensory, and tactile) in arm reaching tasks. To understand the process of sensorimotor integration during skill acquisition in these tasks, we perform measurements of brain activity and movement performance and also develop a model of sensorimotor integration. This project is conducted in collaboration with Drs. Zhigang Zhu, Tony Ro, Ying Li Tian, Kok-Meng Lee and Lewis Wheaton.

- Development of Tongue-Operated Exoskeleton for Stroke Rehabilitation

In this project we collaborate with the groups of Drs. Maysam Ghovanloo (Georgia Tech), Andrew J. Butler (Georgia State University), and Minoru Shinohara (Georgia Tech) to develop novel therapeutic options for people with severe motor disabilities of the upper limbs.
 Mechanics and Control of Precise Stepping
Mechanics and Control of Precise Stepping
In this collaborative project with Dr. Beloozerova (Barrow Neurological Institute) we ask how accurate stepping movements are organized and achieved and what are the biomechanical and neural mechanisms of accurate locomotor movements. We address these questions by assessing full-body mechanics, limb muscle activity, and activity of motor cortex during locomotor task with different demands on the accuracy of foot placement. Our subjects walk on a continuous flat surface, on horizontal ladders with rungs of different widths, and along narrow pathways.
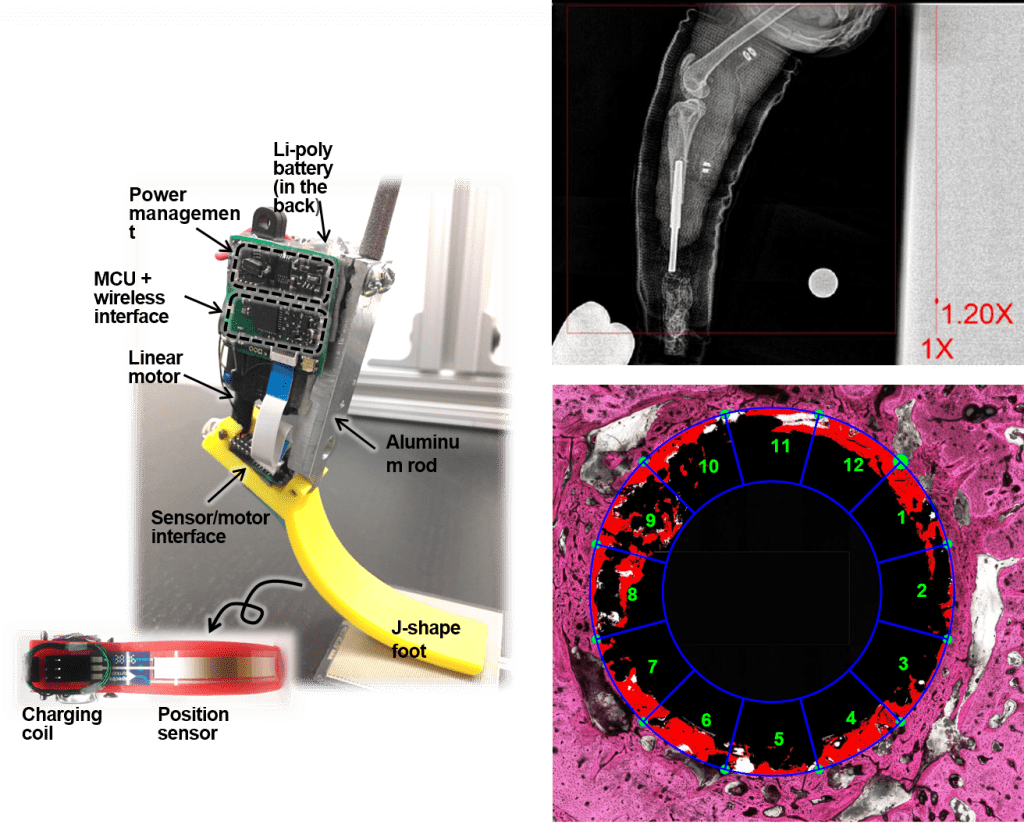
- Integration of the residual limb with a prosthesis via direct skin-bone-peripheral nerve interface. This collaborative project with Drs. Mark Pitkin (Tufts University), Hangue Park (Taxes A&M University), Stephen P. DeWeerth (Lehigh University), and Martha Grover (Georgia Tech) focuses on the development of a sensing and powered limb prosthesis integrated with skin, bone and peripheral nerves and muscles.
Research Opportunities
Research opportunities related to the projects listed above are available for graduate and undergraduate students. Dr. Prilutsky is also a faculty member of the Georgia Tech’s Bioengineering Graduate Program and an adjunct faculty member of the Division of Physical Therapy at Emory University School of Medicine. Undergraduate students interested in research opportunities in the lab are encouraged to apply for the the President’s Undergraduate Research Awards.
Selected Publications
- Books and book chapters
- Prilutsky BI, Zatsiorsky VM. Neural Control Principles: Bernstein’s Insights from Biomechanics of Human Movement. In: Bernstein’s Construction of Movements: The Original Text and Commentaries, edited by Latash ML. New York and London: Routledge, p. 272-285, 2021. PDF.
- Edwards, D.H., Prilutsky, B.I. Sensory feedback in the control of posture and locomotion. In: Neurobiology of Motor Control: Fundamental Concepts and New Directions (eds. Hooper SL, Büschges A). New York: Wiley, 263-304, 2017. PDF.
- Prilutsky BI, Edwards DH (editors). Neuromechanical Modeling of Posture and Locomotion. New York: Springer, 2016.
- Bondy B, Klishko AN, Edwards DH, Prilutsky BI, Cymbalyuk G. Control of cat walking and paw-shake by a multifunctional central pattern generator. In: Neuromechanical Modeling of Posture and Locomotion (eds. Prilutsky BI, Edwards DH, Jr). New York, NY: Springer, p. 333-359, 2016. PDF.
- Markin SN, Klishko AN, Shevtsova NA, Lemay MA, Prilutsky BI, Rybak IA. A neuromechanical model of spinal control of locomotion. In: Neuromechanical Modeling of Posture and Locomotion (eds. Prilutsky BI, Edwards DH, Jr). New York, NY: Springer, p. 21-65, 2016. PDF.
- Prilutsky BI, Klishko AN, Weber DJ, and Lemay MA. Computing motion dependent afferent activity during cat locomotion using a forward dynamics musculoskeletal model. In: Neuromechanical Modeling of Posture and Locomotion (eds. Prilutsky BI, Edwards DH, Jr). New York, NY: Springer, p. 273-307, 2016. PDF.
- Shevtsova NA, Hamade K, Chakrabarty S, Markin SN, Prilutsky BI, and Rybak IA. Modeling the organization of spinal neural circuits controlling two-joint muscles In: Neuromechanical Modeling of Posture and Locomotion (eds. Prilutsky BI, Edwards DH, Jr). New York, NY: Springer, p. 121-162, 2016. PDF.
- Zatsiorsky VM and Prilutsky BI. Biomechanics of Skeletal Muscles. Champaign, IL: Human Kinetics, 2012. (Japanese translation).
- Selected peer-reviewed articles
- Prilutsky BI, Aruin AS (2023) Contributions of Vladimir M. Zatsiorsky to human movement science. Hum Mov Sci 87:103048. PDF.
- Audet J, Harnie J, Lecomte CG, Mari S, Merlet AN, Prilutsky BI, Rybak IA, Frigon A. Control of Forelimb and Hindlimb Movements and Their Coordination during Quadrupedal Locomotion across Speeds in Adult Spinal Cats. J Neurotrauma 39:1113-1131, 2022. PDF.
- Park H, Klishko AN, Oh K, Zhang C, Grenga G, Herrin KR, Dalton JF, Kistenberg RS, Lemay MA, Pitkin M, DeWeerth SP, Prilutsky BI. Electrical Stimulation of Distal Tibial Nerve During Stance Phase of Walking May Reverse Effects of Unilateral Paw Pad Anesthesia in the Cat. Motor Control: 1-25, 2022. PubMed
- Oh K, Prilutsky BI. Transformation from arm joint coordinates to hand external coordinates explains non-uniform precision of hand position sense in horizontal workspace. Hum Mov Sci 86:103020, 2022. PDF.
- Prilutsky BI, Parker J, Cymbalyuk GS, Klishko AN. Emergence of Extreme Paw Accelerations During Cat Paw Shaking: Interactions of Spinal Central Pattern Generator, Hindlimb Mechanics and Muscle Length-Depended Feedback. Front Integr Neurosci 16:810139, 2022. PubMed
- Frigon A, Akay T, and Prilutsky BI. Control of mammalian locomotion by somatosensory feedback. Compr Physiol 12: 2877-2947, 2021. PubMed
- Parker JR, Klishko AN, Prilutsky BI, and Cymbalyuk GS. Asymmetric and transient properties of reciprocal activity of antagonists during the paw-shake response in the cat. PLoS Comput Biol 17: e1009677, 2021. PubMed
- Zhang Z, Prilutsky BI, Butler AJ, Shinohara M, Ghovanloo M. Design and preliminary evaluation of a tongue-operated exoskeleton system for upper limb rehabilitation. Int J Environ Res Public Health, 18(16): 8708, 2021. PubMed
- Klishko AN, Akyildiz A, Mehta-Desai R, Prilutsky BI. Common and distinct muscle synergies during level and slope locomotion in the cat. J Neurophysiol., 126(2): 493-515, 2021. PubMed
- Pitkin M, Cassidy C, Shevtsov MA, Jarrell JR, Park H, Farrell BJ, Dalton JF, Childers WL, Kistenberg RS, Oh K, Klishko AN, Prilutsky BI. Recent Progress in animal studies of the skin- and bone-integrated pylon with deep porosity for bone-anchored limb prosthetics with and without neural interface. Mil Med. 186(Suppl 1):688-695, 2021. PubMed
- Harnie J, Audet J, Klishko AN, Doelman A, Prilutsky BI, Frigon A. The spinal control of backward locomotion. J Neurosci. 41(4):630-647, 2021. PubMed
- Latash EM, Barnett WH, Park H, Rider JM, Klishko AN, Prilutsky BI, Molkov YI. Frontal plane dynamics of the centre of mass during quadrupedal locomotion on a split-belt treadmill. J R Soc Interface 17(170):20200547, 2020. PubMed
- Higgin D, Krupka A, Maghsoudi OH, Klishko AN, Nichols TR, Lyle MA, Prilutsky BI, Lemay MA. Adaptation to slope in locomotor-trained spinal cats with intact and self-reinnervated lateral gastrocnemius and soleus muscles. J Neurophysiol. 123(1):70-89, 2020. PubMed
- Park H, Latash EM, Molkov YI, Klishko AN, Frigon A, DeWeerth SP, Prilutsky BI. Cutaneous sensory feedback from paw pads affects lateral balance control during split-belt locomotion in the cat. J Exp Biol 222(Pt 14). pii: jeb198648. doi: 10.1242/jeb.198648, 2019. PubMed
