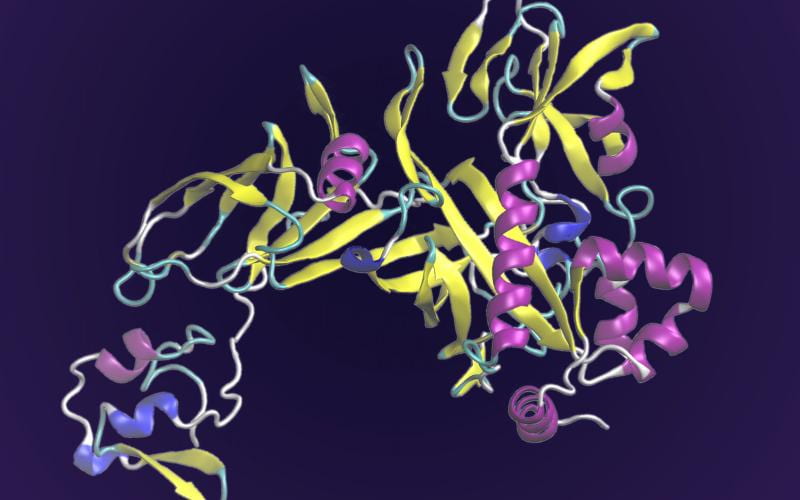
Welcome to the Center for the Study of Systems Biology at Georgia Tech

What is Systems Biology?
Recognized by most experts in the field as the future of biology, Systems Biology seeks to understand how complex living systems interact with each other so that we can diagnose and treat disorders such as cancer. While past biological research has taught us much about how these individual biological units are structured and function, future biology will be focused on understanding how these units interact.
Vision of the Center for the Study of Systems Biology
With the completion of the sequencing of the human genome, the possibility exists to unlock the secrets of life and with such understanding, powerful new approaches to the treatment of disease will emerge. Read More →
Research Highlights
COVER: Journal of Chemical Information and Modeling
Entabolons: How Metabolites Modify the Biochemical Function of Proteins and Cause the Correlated Behavior of Proteins in Pathways
Although there are over 100,000 distinct human metabolites, their biological significance is often not fully appreciated. Metabolites can reshape the protein pockets to which they bind by COLIG formation, thereby influencing enzyme kinetics and altering the monomer–multimer equilibrium in protein complexes. Binding a common metabolite to a set of protein monomers or multimers results in metabolic entanglements that couple the conformational states and functions of nonhomologous, nonphysically interacting proteins that bind the same metabolite. These shared metabolites might provide the collective behavior responsible for protein pathway formation. Proteins whose binding and functional behavior is modified by a set of metabolites are termed an “entabolon”─a portmanteau of metabolic entanglement and metabolon. 55%–60% (22%–24%) of pairs of nonenzymatic proteins that likely bind the same metabolite have a p-value that they are in the same pathway, which is <0.05 (0.0005). Interestingly, the most populated pairs of proteins common to multiple pathways bind ancient metabolites. Similarly, we suggest how metabolites can possibly activate, terminate, or preclude transcription and other nucleic acid functions and may facilitate or inhibit the binding of nucleic acids to proteins, thereby influencing transcription and translation processes. Consequently, metabolites likely play a critical role in the organization and function of biological systems.
 New AI Tool Identifies Better Antibody Therapies
New AI Tool Identifies Better Antibody Therapies
From sending cancer into remission to alleviating Covid-19 symptoms, immunotherapy can provide revolutionary disease treatments. Immunotherapies use antibodies — proteins that bind to cell markers called antigens — to target and eliminate the antigen. But despite how effective immunotherapy can be, it isn’t widely used because finding the right antibodies to develop treatments is challenging, time-consuming work.
Georgia Tech researchers are making this process a little easier, though. Their new tool, AF2Complex, used deep learning to predict which antibodies could bind to Covid-19’s infamous spike protein. The researchers created input data for the deep-learning model using sequences of known antigen binders. This method correctly predicted 90% of the best antibodies in one test with 1,000 antibodies and was recently published in Proceedings of the National Academy of Sciences. Treating Covid-19 is just the start of its potential.
 Trial Begins For ‘Probabilistic Approach’ To Diagnosing Ovarian Cancer
Trial Begins For ‘Probabilistic Approach’ To Diagnosing Ovarian Cancer
Scientists in the Georgia Tech Integrated Cancer Research Center (ICRC) are proposing a new, more realistic approach to diagnosing cancer that provides a probabilistic statement about the likelihood of developing it, much like how cholesterol tests are used to assess heart disease risk. Probabilities are based on an individual’s metabolic profile and the first population group to be targeted are women at high risk for ovarian cancer, according to John McDonald, Ph.D., professor emeritus in the school of biological sciences at Georgia Institute of Technology and founding director of the ICRC.
*Also featured on Clinical Research News.

Researchers Leverage AI to Develop Early Diagnostic Test for Ovarian Cancer
For over three decades, a highly accurate early diagnostic test for ovarian cancer has eluded physicians. Now, scientists in the Georgia Tech Integrated Cancer Research Center (ICRC) have combined machine learning with information on blood metabolites to develop a new test able to detect ovarian cancer with 93 percent accuracy among samples from the team’s study group.
*Also featured on the New York Post, Atlanta New First, Medical Xpress, News Medical Life Sciences, Medscape, AJMC, ScienceDaily, Life Technology, Omics Tutorials, Oncology News Australia, Newswise and Eurekalert.
 The University System of Georgia (USG) Board of Regents has affirmed the renewal of Jeffrey Skolnick as a Regents’ Professor!
The University System of Georgia (USG) Board of Regents has affirmed the renewal of Jeffrey Skolnick as a Regents’ Professor!
Regents’ distinctions may be granted for a period of three years by the Board of Regents (BOR) to outstanding faculty members from Georgia Tech, Augusta University, Georgia State University, the University of Georgia, and, in special circumstances, other USG institutions. A Regents’ professor, researcher, or entrepreneur distinction is awarded only after unanimous recommendation from the president of the recipient’s university, their chief academic officer and dean, as well as three additional members of the faculty who are named by the university president.
Mu Gao wins the 2023 Outstanding Sr. Research Faculty Award!
Mu Gao was awarded with the 2023 Outstanding Sr. Research Faculty Award by the Georgia Tech College of Sciences at the Spring Science Celebration, Celebrating Excellence in Research, Teaching, and Community hosted by Susan Lozier, Dean of the College of Sciences and Betsy Middleton and john Clark Sutherland Chair.
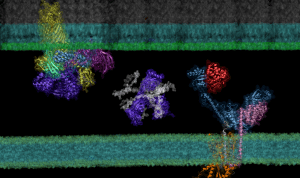 AF2Complex ‘Computational Microscope’ Predicts Protein Interactions, Potential Paths to New Antibiotics
AF2Complex ‘Computational Microscope’ Predicts Protein Interactions, Potential Paths to New Antibiotics
Though it is a cornerstone of virtually every process that occurs in living organisms, the proper folding and transport of biological proteins is a notoriously difficult and time-consuming process to experimentally study.
 Computing function from form
Computing function from form
Georgia Tech and Oak Ridge National Laboratory researchers extend AI’s prediction prowess to complex protein structures.
Over the past two years, artificial intelligence has shown it can predict what many cellular components look like. For instance, the AlphaFold deep-learning tool developed by Google sister company DeepMind has decoded how nearly every amino acid sequence folds into defined shapes.

AF2Complex: Researchers Leverage Deep Learning to Predict Physical Interactions of Protein Complexes
A computational tool developed to predict the structure of protein complexes – the molecular machinery that makes life possible – is lending new insights into the biomolecular mechanisms of their function.
Scientists use Summit supercomputer, deep learning to predict protein functions at genome scale
A team of scientists led by the Department of Energy’s Oak Ridge National Laboratory and the Georgia Institute of Technology is using supercomputing and revolutionary deep learning tools to predict the structures and roles of thousands of proteins with unknown functions.

Georgia Tech researchers making breakthroughs treating COVID-19
WSB-TV Channel 2 Action News coverage of MOATAI-VIR.
 Emory, Georgia Tech researchers develop AI-tool to predict COVID-19 symptoms
Emory, Georgia Tech researchers develop AI-tool to predict COVID-19 symptoms
Researchers at Emory and the Georgia Institute of Technology (Georgia Tech) published a paper on Oct. 21 detailing an artificial intelligence-based tool that is able to predict COVID-19 symptoms and suggest specific FDA-approved drugs.
AI Tool Pairs Protein Pathways with Clinical Side Effects, Patient Comorbidities to Suggest Targeted Covid-19 Treatments
The symptoms and side effects of Covid-19 are scattered across a diagnostic spectrum. Some patients are asymptomatic or experience a mild immune response, while others report significant long-term illnesses, lasting complications, or suffer fatal outcomes.
Three researchers from the Georgia Institute of Technology and one from Emory University are trying to help clinicians sort through these factors and spectrum of patient outcomes by equipping healthcare professionals with a new “decision prioritization tool.” PDF

Jeffrey Skolnick, Andre Ghetti (ANABIOS Corporation), Nicole Jung (Karlsruhe Institute of Technology) and Hongyi Zhou win the 2020 NCATS ASPIRE Reduction-to-Practice Challenge, Stage 2, Milestone 1: Prototype Development and Milestones Delivery for their application entitled “Development of a Comprehensive Integrated Platform for Translational Innovation in Pain, Opioid Abuse Disorder and Overdose”
The Reduction-to-Practice Challenge consists of three stages: (1) Planning (winners announced in April 2021); (2) Prototype Development and Milestone Delivery; and (3) Prototype Delivery, Independent Validation and Testing. This stage of the challenge was the Prototype development.
 ASPIRE-ing To Find Fast Solutions to the Opioid Health Crisis
ASPIRE-ing To Find Fast Solutions to the Opioid Health Crisis
School of Biological Sciences’ Jeffrey Skolnick and Hongyi Zhou are part of an award-winning NIH effort to create innovative, AI-powered platforms for discovering new pain management drugs — and identify immediate solutions.
 Jeffrey Skolnick, Andre Ghetti (ANABIOS Corporation), Nicole Jung (Karlsruhe Institute of Technology) and Hongyi Zhou win the 2020 NCATS ASPIRE Reduction-to-Practice Challenge for their application entitled “Development of a Comprehensive Integrated Platform for Translational Innovation in Pain, Opioid Abuse Disorder and Overdose”
Jeffrey Skolnick, Andre Ghetti (ANABIOS Corporation), Nicole Jung (Karlsruhe Institute of Technology) and Hongyi Zhou win the 2020 NCATS ASPIRE Reduction-to-Practice Challenge for their application entitled “Development of a Comprehensive Integrated Platform for Translational Innovation in Pain, Opioid Abuse Disorder and Overdose”
The goal of this Challenge is to combine the best solutions and develop a working platform that integrates the four component areas. The Reduction-to-Practice Challenge consists of three stages: (1) Planning; (2) Prototype Development and Milestone Delivery; and (3) Prototype Delivery, Independent Validation and Testing. The first stage of this Challenge required the submission of a plan for the reduction-to-practice of a platform that integrates the four component areas into a comprehensive solution.
MOATAI-VIR: Hot Topic of the Day on 02/02/2021 on the Centers for Disease Control and Prevention, Public Health Genomics and Precision Health Knowledge Base webpage.
Astore, C, Zhou H, Jacob J, Skolnick J. 2021. MOATAI-VIR – an AI algorithm that predicts severe adverse events and molecular features for COVID-19’s complications. medRxiv 2021.01.29.21250712; doi: https://doi.org/10.1101/2021.01.29.21250712. PDF
 Jeffrey Skolnick Receives Regents Recognition
Jeffrey Skolnick Receives Regents Recognition
Seven Georgia Tech Faculty Members Receive Regents Recognition – The University System of Georgia (USG) Board of Regents (BOR) appointed seven Georgia Tech faculty members Regents Professors, the highest academic recognition bestowed by the USG. The seven Regents Professors are:
- Marilyn Brown, Brook Byers Professor in Sustainable Manufacturing in the School of Public Policy
- Suresh Sitaraman, Morris M. Bryan Jr. Professor in Mechanical Engineering in the George W. Woodruff School of Mechanical Engineering
- Jeffrey Skolnick, Mary and Maisie Gibson Chair in Computational Systems Biology and GRA Eminent Scholar in the School of Biological Sciences
- Prasad Tetali, professor in the School of Mathematics and the School of Computer Science
- Vigor Yang, professor in the Daniel Guggenheim School of Aerospace Engineering
- Lisa Yaszek, professor in the School of Literature, Media, and Communication
- Ellen Zegura, Stephen Fleming Chair in the School of Computer Science
 Origin of Life’s Handedness and Protein Biochemistry
Origin of Life’s Handedness and Protein Biochemistry
Examine your hands. The right is a mirror image of the left. They look very similar, but you know they’re not when you try to put your left hand inside a right glove.
The molecules of life have a similar handedness. Proteins for example are like your left hand, made up of amino acids that are all left-handed. This phenomenon is called chirality. How chiral systems emerged is one of the key questions of origins-of-life research.
 On the possible origin of protein homochirality, structure, and biochemical function
On the possible origin of protein homochirality, structure, and biochemical function
Living systems contain mainly chiral macromolecules, including proteins. How L-chiral proteins emerged from demi-chiral mixtures is unknown. Our simulations show that, compared to contemporary proteins, demi-chiral proteins have shorter regular secondary structures due to fewer internal hydrogen bonds, but similar global folds and small molecule binding sites. Demi-chiral proteins contain L-chiral substructures matching native active site geometries. Among the most frequently generated enzymes with native active site residues are ancient functions associated with metabolism and replication. This suggests that demi-chiral proteins could engage in early metabolism, creating the feedback loop for transcription and cell formation partly responsible for life’s emergence.